| Issue |
BIO Web Conf.
Volume 30, 2021
II International Symposium “Innovations in Life Sciences” (ILS 2020)
|
|
|---|---|---|
| Article Number | 04001 | |
| Number of page(s) | 3 | |
| Section | Ecological Questions, Environmental Issues and Environmental Education in Modern World | |
| DOI | https://doi.org/10.1051/bioconf/20213004001 | |
| Published online | 22 April 2021 | |
Mobility of сarbohydrates in tulip bulbs
1
Department of Biology Belgorod State National Research University, Belgorod, Russia
2
Department of Foreign Languages and Professional Communication Belgorod State National Research University, Belgorod, Russia
3
Department of Сhemistry Belgorod State National Research University, Belgorod, Russia
Abstract
Quantitative determination of carbohydrates in Tulip bulbs by spectrophotometry using a method that allows determining the content of monosaccharides, soluble oligosaccharides and starch in a single sample was conducted. The mobility of carbohydrates within the Tulip bulb in conditions of forcing and growing in the open ground is demonstrated.
© The Authors, published by EDP Sciences, 2021
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
1 Introduction
It is known that bulbs contain 7-21 % of dry matter, including 4-16 % of carbohydrates. Among them, there are monosaccharides and oligosaccharides and polysaccharides formed from them during polymerization [1]. Carbohydrates are the primary products of biosynthesis. They are formed during photosynthesis and they act as the main nutrient and main support material for plant cells and tissues. Carbohydrates are involved in many metabolic processes, but they are primarily the main energy suppliers [2]. Spare carbohydrates can cross over into each other [3, 4, 5, 6]. The main reserve carbohydrate is starch, which decomposes under the action of a enzymes-amylases group and with the participation of hormones. Glucose and other monosaccharides are known to be the main sources of energy. Sucrose and other oligosaccharides act as intermediate compounds and play an important role in regulating plant growth and development [6]. These transformations have a strict dependence on morphological, anatomical and physiological transformations [3, 7, 8]. Variability of carbohydrate and hormonal status of bulb tissues and its influence on the number of flowers is shown [3].
It is known that in varietal tulips, temperature fluctuations affect the ratio of various carbohydrates, and under unfavorable conditions, rapid depletion of energy resources of the tissues storing scales can occur [8].
These changes require energy and plastic substances. The synchronous nature of transformations is of both scientific and practical interest. Simultaneous determination of different forms of carbohydrates in plant material allows us to establish the regularities of these substances metabolism at different stages of ontogenesis. The purpose of our study is to demonstrate the change in the content of various carbohydrates of bulbs when growing in open and protected dirt from the bulbs that have passed the same dormant period, when all the elements of the generative shoot and flower were laid.
2 Experimantal
The mobility of spare carbohydrates in Tulip bulbs during distillation and cultivation in the open dirt (the garden) was studied in the laboratory using the method of determining the content of water-soluble sugars and starch from a single sample [9]. The object of the study was Tulip bulbs of the “Parad” variety, the class of “Darwin hybrids”. The bulbs were planted in the open dirt in October, when the average daily temperature was 6-9 °C [10]. For distillation, the bulbs were placed in the refrigerator for cooling at a temperature of 4-6 °C at the end of September. Bulbs were planted for rooting on November 14, and were brought into the light on January 20 [9]. For biochemical studies, 10 bulbs were taken in the main stages of nutrient consumption: the first monitoring was held during the rooting of the bulbs, the second monitoring was held during the growth of the green mass. Storage scales were extracted from the bulbs, crushed, and monosaccharides, soluble oligosaccharides, and starch were determined in a pattern taken from a single sample [11].
Determination of the sugar content was carried out in the outer storage scales of the bulb (1), intermediate scales of the bulb (2) and the sprout and adjacent scales of the bulb (3). According to the method [9], the sample attachments were crushed and filled with hot distilled water. By centrifugation, the settling liquid was separated for further determination of water-soluble sugars. The sediment was subjected to acid treatment for the purpose of hydrolysis of the starch contained in it. The concentration of carbohydrates was determined spectrophotometrically by changing the colour intensity of the products of interaction of picric acid with carbohydrates in an alkaline medium when heated. Statistical processing of the obtained results was performed using a standard Microsoft Excel program.
3 Results ans discussion
When vegetation ends, the life of the bulbous plant does not stop. During the dormant period in the daughter bulb, the future shoot is formed (Fig. 1), and the future flower is differentiated from the growth cone in late July and early August. At the same time, another renewal bud is laid at the base of the peduncle, that is, the future replacement bulb, as well as daughter bulbs. The formation of the future flower organs is completed in September-October. At this time, the effect of low positive temperatures is very important. Since July, new adventitious roots are laid in the tissue of the Donets of a young bulb start active growth which in conditions of temperature decrease from +9 °C [12]. They will provide the plant with the necessary water and minerals in the next growing season [13].
The study found that the content of monosaccharides in different parts of the bulb is different and more in the outer part of the bulb. In the external storage scales, they are determined from 1.6 mg/g to 7.6 mg/g, while in the sprout and adjacent scales of the bulb from 1.1 mg/g to 5.4 mg/g. Data indicate a greater presence of monosaccharides in all parts of the bulb during green mass growth than during rooting (Table I).
Comparison of the study results according to the method of cultivation shows that at the time of the first monitoring (rooting of bulbs) a greater content of monosaccharides in the outer and intermediate storage scales of bulbs was determined twice as much as when growing in the garden. The conditions for keeping plants during this period are very different. In protected dirt, humidity, soil temperature and air do not change much. In open dirt conditions, these indicators can vary greatly, which determines the different intensity of metabolism. It is known that carbohydrates can perform cryoprotective and signaling functions. Thus, maltodextrins, being soluble oligosaccharides, are able to preserve the protein structure and reduce the effect of toxins when cooled [14]. Sugars can influence the expression of the nuclear genome and organelle genomes no less strongly than hormones [15]. The ratio of various carbohydrates leads to a redistribution of transport flows between the root and stem apexes and transfers the plant to the generative stage [16]. And certain ratios of temperature and light change the direction of carbohydrates and hormones influence on the plant genome [17, 18]. Probably the monosaccharides are consumed by the bulbs for protection of generative shoots. However, at the time of the second monitoring (green mass growth) the content of monosaccharides in all parts of Tulip bulbs during distillation is 1.7-1.8 times higher than in bulbs grown in the open dirt. Thus, growing tulips in protected dirt is a more energyconsuming process at the stage of vegetative growth.
Water-soluble polysaccharides play the role of available reserve carbohydrates. Typical and most common representatives of plant oligosaccharides are oligosaccharides of the sucrose group: melezitose, raffinose, gentians, stachyose, etc. The content of watersoluble polysaccharides when rooting is five or more times higher in the garden in all parts of the bulb (Table II). Probably it is connected with the unstable conditions and the expenditure of oligosaccharides to support life processes through changes in metabolism. Besides, the root system in the conditions of distillation is less developed, therefore, less plastic and energy substances are required for its formation. At the same time, there are fewer oligosaccharides in the sprout and the adjacent scales of the bulb.
Starch, as the main substance of long-term storage, is contained from 0.1 mg/g in the average scale at the stage of green mass growth in the closed dirt to 3.6-4 mg / g at the stage of rooting bulbs (Table III). At the rooting stage, the starch bulbs are larger under different growing conditions in different parts, with the exception of the open dirt. There is almost seven times more starch here. When building up the green mass of starch is more in the open dirt.
According to the results of our studies, which are presented graphically (Fig. 2), in the conditions of protected dirt, a higher content of starch in all parts of the bulbs was found, there are significantly less monosaccharides and even less oligosaccharides. However, at the time of the second monitoring, more starch is contained in bulbs grown in the open dirt. During the growing season, starch is consumed in all parts of the bulb with different intensity. At this time, the number of oligosaccharides increases by 6-13 times, and by 5-6 times for monosaccharides.
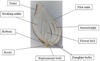 |
Fig. 1. Tulip Bulb Structure |
The Content Of Monosaccharides In Tulip Bulbs (mg/g)
The content of water-soluble polysaccharides in tulip bulbs (mg/g)
The starch content in tulip bulbs (mg / g)
 |
Fig. 2. Dynamics of carbohydrate content: A-forcing, B-the garden 1 – first scale, 2 – second scale, 3 – flower bud |
4 Conclusion
In open dirt conditions, all parts of the bulb contain a more equal content of various types of carbohydrates. It is necessary to note the movement of all carbohydrates from the outer and intermediate storage scales of the bulb during the growing season to the sprout, where implements the generative phase of this season realizes and the basis for future development is laid.
References
- Ye.S. Severin, Biokhimiya, (GEOTAR-MED, Moscow, 2003) [Google Scholar]
- B.P. Pleshkov, Biochemistry of Agricultural Plants, Moscow 496 p.: (2013). [Google Scholar]
- V.V. Kondratyev, M.V. Semenova, T.V. Voronkova, N.N. Danilina. Plant Physiology, 56 (3), 471–478 (2009) [Google Scholar]
- G.-V. Heldt Plant Biochemistry. (2014) [Google Scholar]
- Shelepova, T.V. Voronkova, V.V. Kondratiev, N.N. Danilina, Physiology and Biochemistry, cult of plants, 41, 5 (2009). [Google Scholar]
- X.M. Geng, A. Sato, H. Okubo, M. Saniewsk J. Fac. Agr., Kyushu Univ., 52 (2), 315–319 (2007) [Google Scholar]
- M. Saniewski, L. Kawa-Miszczak, E. Wegrzynowicz-Lesiak and H. Okubo, Dormancy in Plants. From Whole Plant Behaviour to Cellular Control, 275-245 (2000) [Google Scholar]
- Y. X. Rong, Effect of Low Temperature on Changes in Endogenous Hormone Level and Plant Development in Lilium and Tulipa, 118 p. (2007) [Google Scholar]
- V.N. Bylov, E.N. Zaitseva, F.M. Zheleznyak, Introduction and culture techniques of flowerdecorative plants, Moscow, p. 96-113. (1977) [Google Scholar]
- Introduction and techniques of culture of flower decorative plants. (Science, Moscow, 1977) [Google Scholar]
- T.V. Voronkova, O.V. Shelepova, «A method for determining the content of water-soluble sugars and starch from a single sample», Patent No. RU 2406293. IPC A01G 7/00. [Google Scholar]
- M. Baranova, «Mnogoletnie travyanistye rasteniya klassa Odnodol’nye v kollekcii otkrytogo grunta Botanicheskogo sada Petra Velikogo BIN RAN», (Rostock HSE St. Petersburg, 2013) [Google Scholar]
- T.N. Glubscheva, O.V. Netsvetaeva, Catalog of tulips of the BelSU Botanical Gardens collection, (CONSTANT, Belgorod, 2011) [Google Scholar]
- T.I. Trunova, Plants and low temperature stress. 64th Timiryazev reading (Science, Moscow, 2007) [Google Scholar]
- Yu.V. Gamaley, The transport system of vascular plants. (SPb, St. Petersburg University, 2004) [Google Scholar]
- G. Bernier, A. Hevelange, C. Houssa et al., Plant Cell. 5. 1147-1155, (1993) [Google Scholar]
- L. Сorbesier, P. Lejeune, G. Bernier, Planta, 206, p. 131-137, (1998) [Google Scholar]
- S.J. Gibson, Plant Physiol, 124, 1532-1539 (2000) [Google Scholar]
- L. Сorbesier, P. Lejeune, G. Bernier, «The role of carbohydrates in the induction of flowering in Arabidopsis thaliana: comparison between the wild type and a starchless mutant», Planta, 206, p. 131137. (1998) [Google Scholar]
- S.J. Gibson, «Plant sugar-response pathways. Part of a complex regulatory web», Plant Physiol, 124, pp. 1532-1539 (2000) [Google Scholar]
All Tables
All Figures
 |
Fig. 1. Tulip Bulb Structure |
| In the text | |
 |
Fig. 2. Dynamics of carbohydrate content: A-forcing, B-the garden 1 – first scale, 2 – second scale, 3 – flower bud |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.



